What Is PGT-P? Preimplantation Genetic Testing for Polygenic Conditions
PGT-P is a type of genetic screening available to parents going through IVF that can affect a future child's genetic predispositions. It can be used for everything from reducing a child's risk of autism to decreasing their risk of getting heart disease, to increasing their IQ.
PGT-P is a significantly more comprehensive version of the sort of genetic testing that has been around for decades and has traditionally been used to screen for monogenic conditions like cystic fibrosis.
You can reach out to us here if you're interested in getting access.
What is PGT-P? How does it work?
Since the early 1990s, parents with a family history of a single gene disorder like Huntington's disease could use IVF to avoid passing that risk on to their child. They could go to a fertility clinic, work with the doctor to make many embryos, then implant an embryo that was unaffected by the disease. This worked!
In countries where this kind of genetic screening was widely adopted, the prevalence of these disorders among newborns declined dramatically. In Israel, where screening and IVF were made free for carriers of cystic fibrosis, the prevalence of the disease among newborns declined by over 90%.
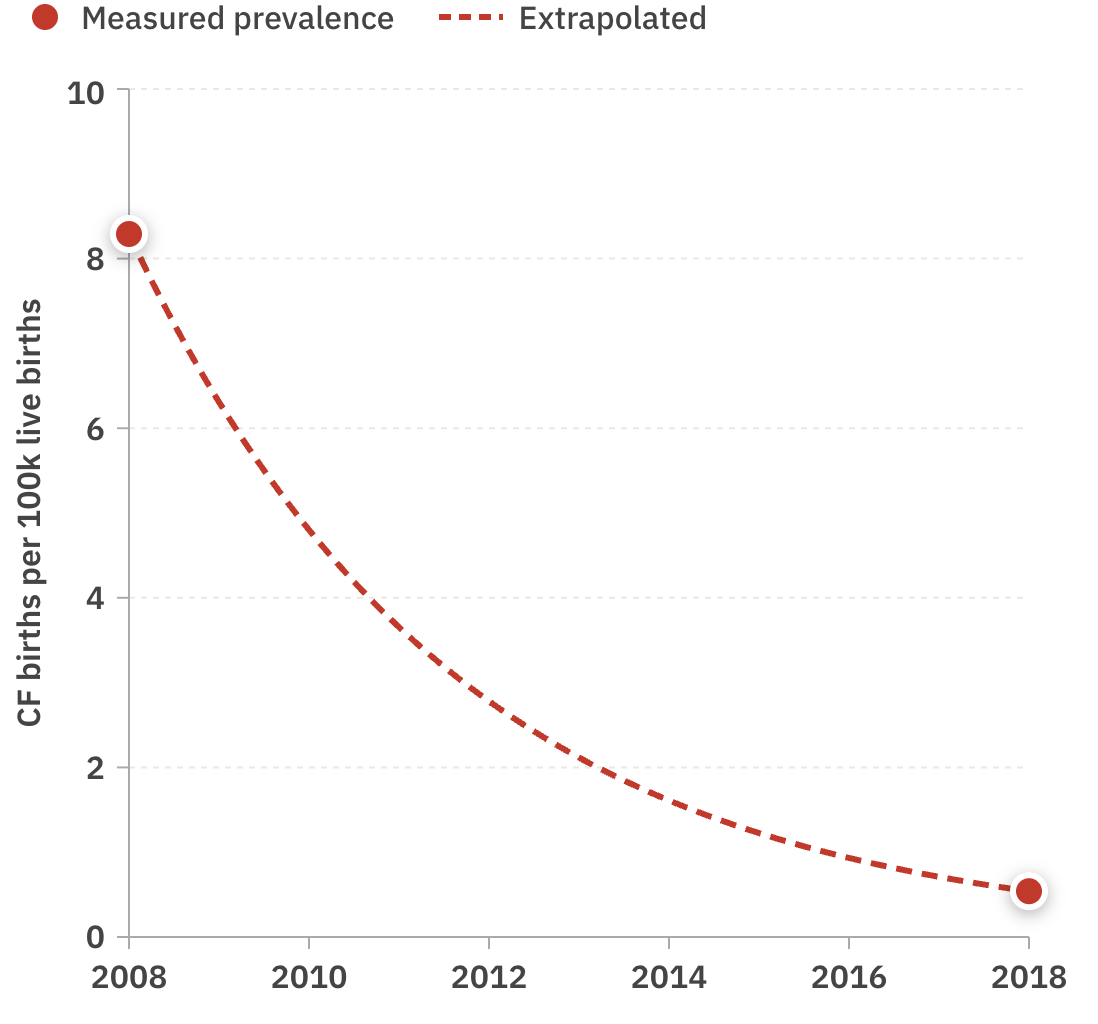
This was a huge victory for public health. But unfortunately most diseases still remained out of reach. Conditions like diabetes, Alzheimer's, heart disease, and schizophrenia still couldn't be tested for.
Since a time before Watson, Crick and Rosalind Franklin unraveled the structure of DNA, we had understood these diseases ran in families. That a large part of whether you developed them was inherited.
But the genetics of these conditions were not as simple as sickle cell or cystic fibrosis. They weren't simply caused by one gene, but many. And no matter how many of these risk genes you carried, they didn't guarantee you would develop it. They simply increased your odds.
In the mid 2010s we finally started to make progress. Researchers began to gain access to giant datasets of genomes and associated health records. For the first time, it became possible to pinpoint individual letters in the DNA that increase someone's risk of a condition like diabetes or schizophrenia.
First dozens, then hundreds, then thousands of genetic variants that affected the risk of all kinds of diseases were uncovered. And the more data we gained access to, the better we began to understand who was at risk and who wasn't.
The formula for calculating someone's risk was fairly simple: first you sequence their DNA, then you look at all the places in their genome that affect their risk of the condition. Some of their genetic variants will increase their risk and some will decrease it. You add up all the effects and compute the person's "polygenic risk score". That number is a summary of their overall risk of getting a disease.
Polygenic risk scores turned out to be quite useful. We found that those with polygenic risk scores in the top 5% for a heart condition like coronary artery disease were over three times as likely to develop the condition as someone with average risk.
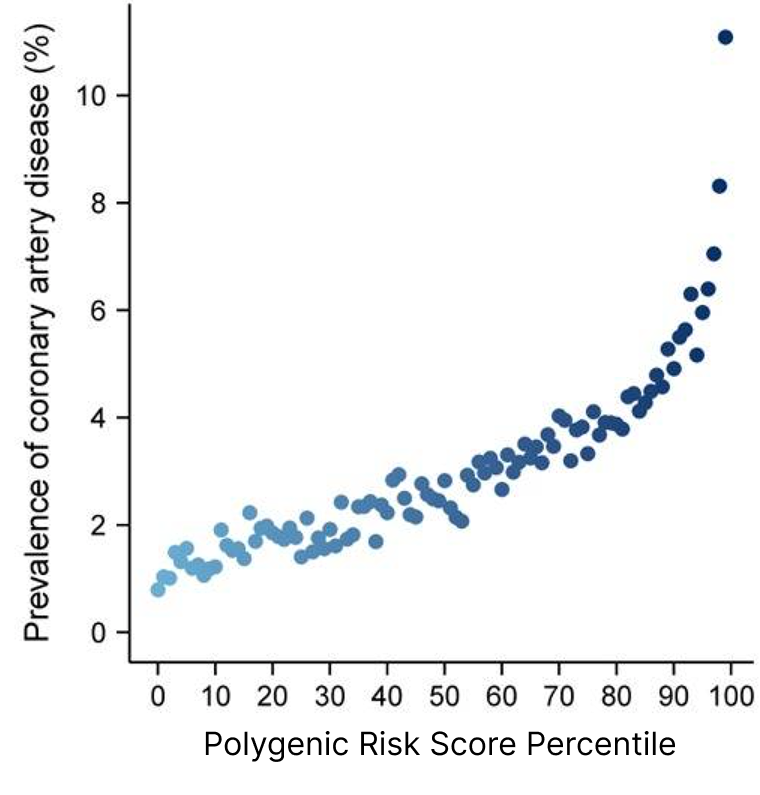
And the better we understood the genes, the bigger these differences became.
Understanding these genes has become quite useful: we now know women with a high risk of breast cancer should start mammograms about a decade earlier than the rest of us. And men with a high risk of prostate cancer could start their PSA tests at a younger age as well.
But perhaps the most intriguing use case for polygenic risk scores was expanding genetic testing of embryos. With this new understanding, we might be able to do to Alzheimer's and diabetes what we've done to cystic fibrosis and sickle cell. All diseases were now, to some degree, preventable.
And diseases were not the only thing on the table. Height, intelligence, even physical appearance are all driven to some degree by genetics. Polygenic scores opened up the possibility of improving a future child's life not just by minimizing downside risk, but also by improving qualities that make for a good life.
The size of the impact you can have on your future child's risk of autism or their IQ or their odds of getting breast cancer varies according to many factors. The two biggest variables are how many embryos you have and how good the predictor is.
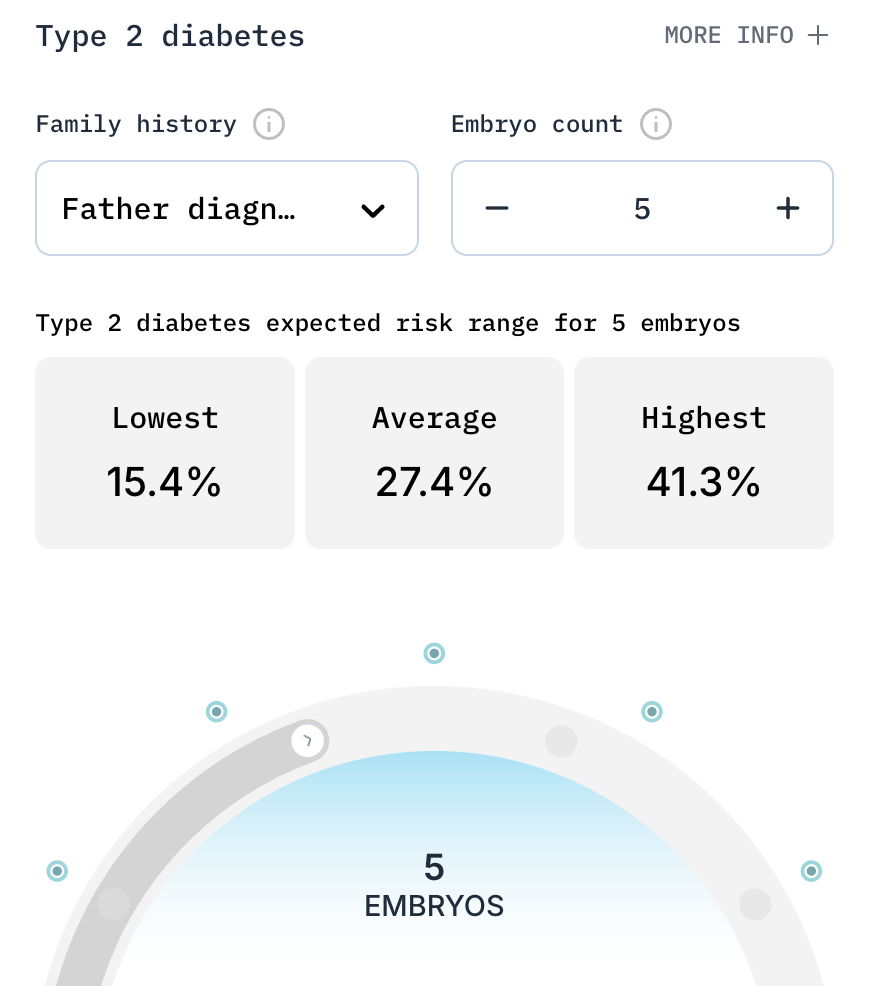
An average 30-year-old couple with five embryos and a father who has type 2 diabetes could reduce their child's risk by an average of 44% just by picking the embryo with the lowest risk.
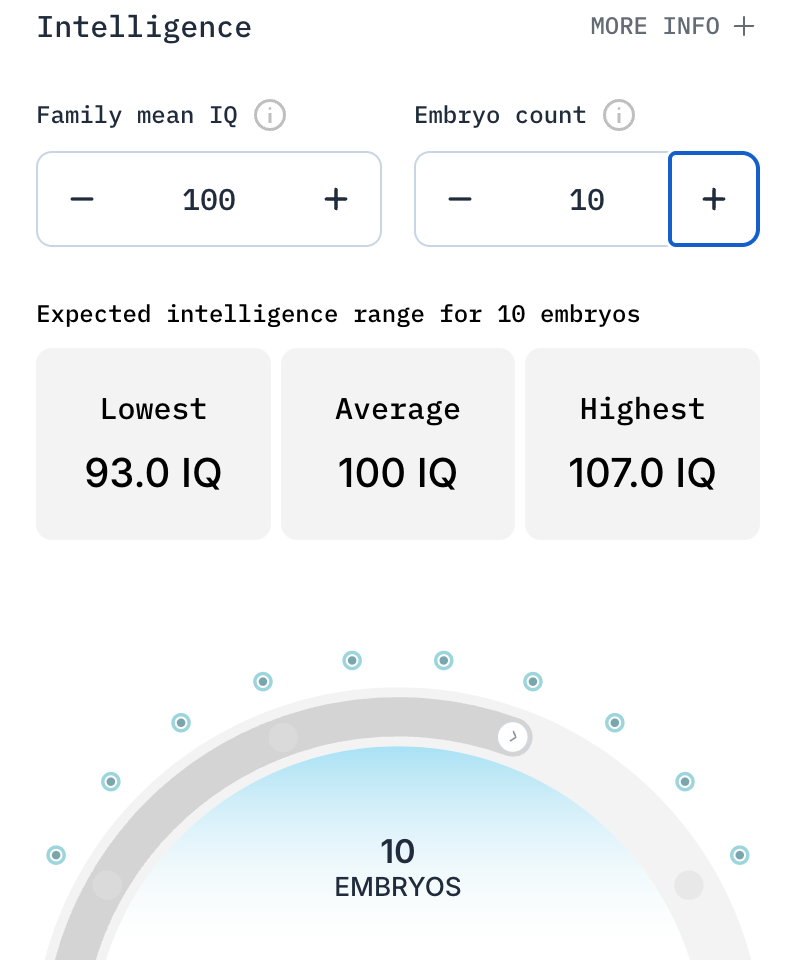
The same couple could increase their child's IQ by about 7 points if they select from the best of 10 embryos.
You can try out the calculator for yourself.
How to get access to PGT-P
If you've done regular PGT-A for your embryos, or if you can get the testing done, Herasight can give you polygenic scores for all of your embryos. A member of our team can walk you through how the process works, what kind of benefits you can get, and pricing. We can even generate a simulated report using the real genomes of you and your spouse to show you how large the benefits would be for your specific genomes.
This can be quite useful because depending on your genetic background the benefits may be larger or smaller than the estimates show.
You can get access by reaching out to us here.
How we know PGT-P works
One might naturally wonder how we can be sure that selecting an embryo with a lower risk of diabetes will actually lower a future child's risk of getting the disease. After all, PGT-P hasn't been around for that long. How do we know it actually works?
The answer is we test these genetic predictors on existing people. We'll train a genetic predictor, then test whether it can predict which people have diabetes.
The gold standard method for testing whether predictors work is called "sibling validation". It's pretty simple: you train a genetic predictor, then you test to see whether it can tell which of two siblings has diabetes, or which one is taller, or which one developed breast cancer.
This method is exceptionally powerful because it proves that the signals detected are actually causing differences rather than just coincidentally associated with it. Each sibling gets a randomized subset of their mother and father's DNA. So if the predictor can tell which siblings are sick, it must be doing so by picking up on real causal signals rather than mere associations.
If you're interested in the science of PGT-P, you can read our validation papers here.